Overview
Diabetes and oxidative stress research laboratory:
The endothelium is the source of a myriad of vasoactive factors that are important for the short and long term regulation of vascular tone, blood flow and the "health" of the cardiovascular system. Unfortunately the endothelium is highly susceptible to damage and this leads to endothelial dysfunction. Metabolic disorders, including insulin resistance and overt diabetes mellitus, are implicated in the development of vascular damage and the acceleration of cardiovascular disease. Endothelial dysfunction is believed to be a critical symptom and an early event leading to cardiovascular disease and complications of diabetes mellitus and is linked to changes in the availability and the balance between the endothelium-derived vasodilatory factor - nitric oxide (NO) and endothelium-derived vaso-constrictors such as endothelin-1 (ET-1).
The primary interest in Dr. Ding's laboratory focuses on the mechanism of diabetes-induced endothelial dysfunction and the contributions of an increase in intracellular oxidative stress linked to hyperglycaemia. We use cell culture system and genetic mouse models of type 2 diabetes to study the cellular basis of endothelial dysfunction in endothelial cells, especially the relationship between alterations in the
bioavailability of NO and an activated endothelin (ET) pathway, linked to increased oxidative stress and changes in the nature/synthesis of other endothelium-derived vasoactive factors. The cause and effect relationship between the increase in oxidative stress in diabetes and the increased activation of NADPH oxidase, shortened telomere length, activation of the ET pathway, and whether any one or all of these diabetes-related changes can be reversed, partially reversed, by therapeutic intervention is also being explored.
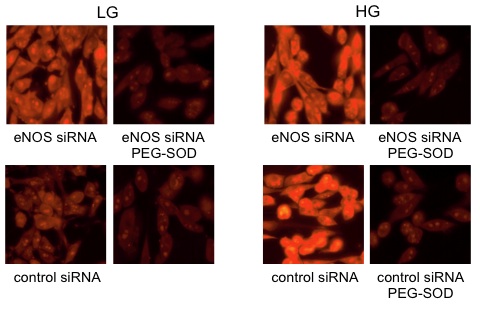
With the use of siRNA against endothelial nitric oxide synthase (eNOS) in a cell culture protocol with mouse microvessel endothelial cells we have demonstrated that in low glucose (LG) conditions eNOS is protective, whereas in high glucose (HG) eNOS contributes to the oxidative stress as measured with dihydroethidium (Aljofan et al., 2007):
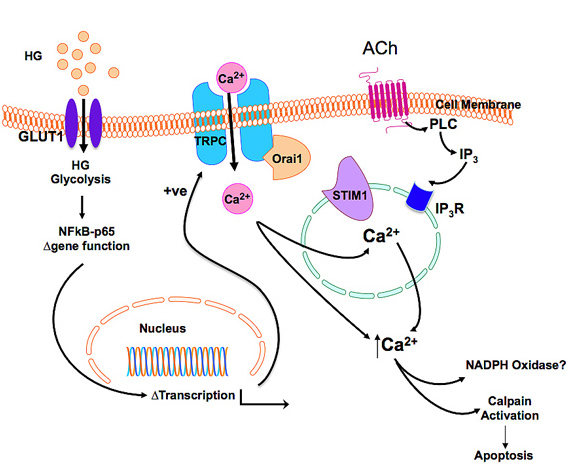
We are also studying the link between hyperglycaemia-induced changes in endothelial cell calcium homeostasis and oxidative stress. Endothelial cells do not express voltage dependent calcium channels and thus agonist-stimulated Ca²+ influx depends on the Ca²+ driving force and the opening of store-operated cation channels (SOCs) by a process referred to as capacitative calcium entry or store-operated calcium entry (SOCE). Changes in Ca²+ homeostasis and eNOS function have been associated with hyperglycaemia and we have provided data that suggests that dysregulation of SOCE, involving transient receptor potential cation channels (TRPC1), contributes to the development of vascular disease (Bishara & Ding, 2010). What is not yet clear is whether changes in TRPC1 expression are linked to the now established role for Stim1 (Stromal Interaction Molecule 1) as the endoplasmic reticulum Ca sensor and Orai1, the pore forming subunit of the highly selective CRAC (Ca²+ release-activated Ca²+ current), in endothelial cells. Published data indicates that TRPC1 co-localises with Orai1 and Stim1 and the exogenous expression of TRPC1 and Stim1 in endothelial cells suggests that Stim1 can regulate TRPC1. We are pursuing studies on the effects of hyperglycaemia on SOCE, changes in Orai1/Stim1 function and the regulation of NADPH oxidase: